Topic Summaries
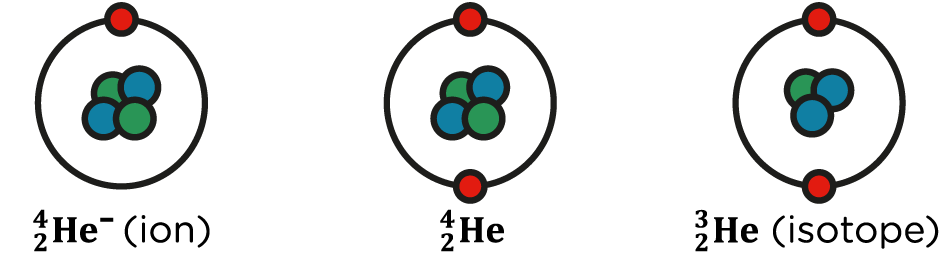
Ions and isotopes
Energy and power
Renewable energy and efficiency
Charge, current, and electric fields
Potential difference and resistors
Electricity
Volume and gases
States of matter and heat capacity
Radiation
Nuclear fusion and fission
Forces
Velocity and acceleration
Gravity and Newton's Laws
Moments and elastic potential
Stopping, braking, and momentum
Properties of waves
Electromagnetic waves
Refraction and lenses
Light, colour, and ray diagrams
Absorbing and emitting infrared radiation
Magnetic fields
Motor effect, generator effect, and transformers
Science skills: Experimental procedures
Science skills: Presenting and using data
Science skills: Measuring results
Energy and power
Renewable energy and efficiency
Charge, current, and electric fields
Potential difference and resistors
Electricity
Volume and gases
States of matter and heat capacity
Radiation
Nuclear fusion and fission
Forces
Velocity and acceleration
Gravity and Newton's Laws
Moments and elastic potential
Stopping, braking, and momentum
Properties of waves
Electromagnetic waves
Refraction and lenses
Light, colour, and ray diagrams
Absorbing and emitting infrared radiation
Magnetic fields
Motor effect, generator effect, and transformers
Science skills: Experimental procedures
Science skills: Presenting and using data
Science skills: Measuring results
Unlock Ions and isotopes
Subscribe to SnapRevise+ to get immediate access to the rest of this resource.
Premium accounts get immediate access to this resource.